Cheetah Medical Announces FDA Clearance and US Release of NIBP Parameter for its NICOM® Hemodynamic Monitoring System
OEM partnership integrates Cheetah Medical’s BIOREACTANCE® noninvasive hemodynamic monitoring core technology with leading SunTech Medical NIBP module to provide advanced hemodynamic monitoring parameters for fluid optimization, goal-directed therapy and patient safety monitoring
Tel-Aviv, Israel and Morrisville, NC, USA, January 7, 2009−Cheetah Medical, a leading provider of fully noninvasive hemodynamic monitoring solutions for the comprehensive care of patients in challenging clinical care settings, has been granted 510(k) marketing clearance by the U.S. Food and Drug Administration (FDA) for the NIBP functionality, provided by SunTech Medical, in its NICOM® Reliant® Hemodynamic Monitoring System.

 press release!aksjnf akjsnfjkasnj kfnasjk fnajsknf jkasnf jkanskjf najksnf jkasnfkj naskjfn kajsnf kjasnfkj naskjfn kjasnfkj naskjnf
press release!aksjnf akjsnfjkasnj kfnasjk fnajsknf jkasnf jkanskjf najksnf jkasnfkj naskjfn kajsnf kjasnfkj naskjfn kjasnfkj naskjnf  Soft Disposable BP Cuffs
Soft Disposable BP Cuffs Neonatal Soft Disposable BP Cuffs
Neonatal Soft Disposable BP Cuffs Vinyl BP Cuffs
Vinyl BP Cuffs All Purpose BP Cuffs
All Purpose BP Cuffs One Piece BP Cuffs
One Piece BP Cuffs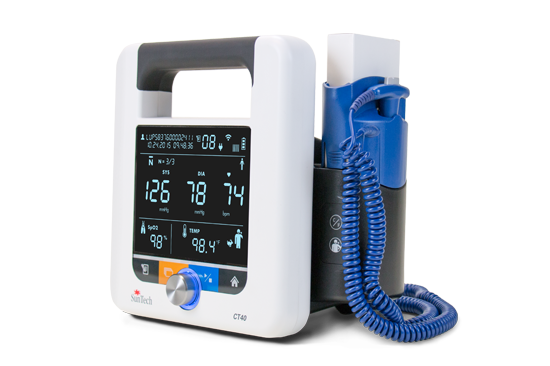 SunTech CT40
SunTech CT40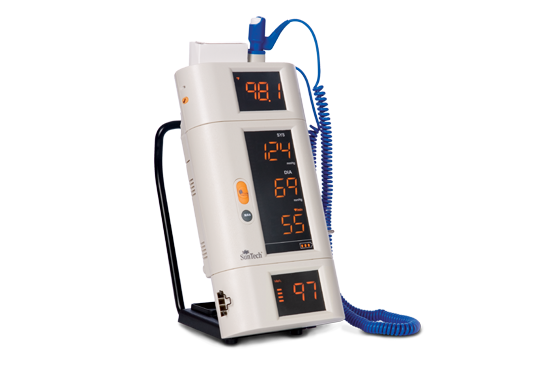 SunTech 247
SunTech 247 Oscar 2
Oscar 2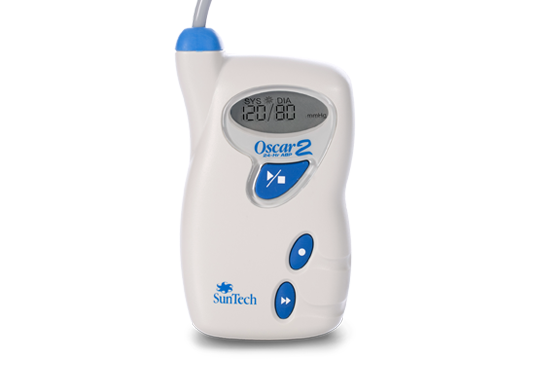 Oscar 2 Classic
Oscar 2 Classic Orbit BP Cuff
Orbit BP Cuff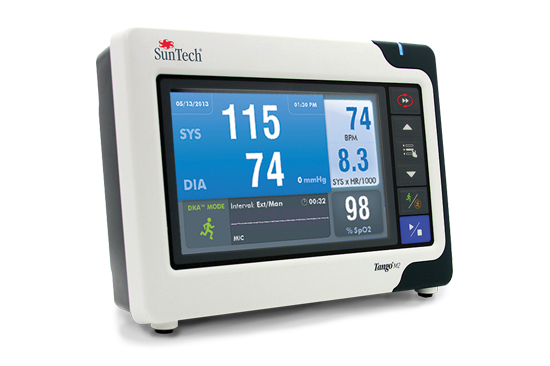 Tango M2
Tango M2 Orbit-K BP Cuff
Orbit-K BP Cuff SunTech Vet20
SunTech Vet20 Vet BP Cuffs
Vet BP Cuffs