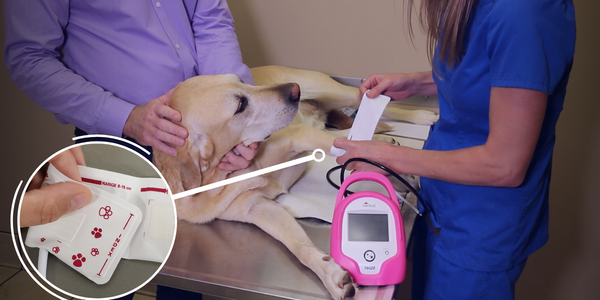
Medical device manufacturers are subject to many rigorous safety standards to ensure that patient’s lives are not endangered due to equipment related errors. One of these standards, ISO 80369-5, establishes a general framework and guidance for small-bore connectors intended for use in healthcare applications, promoting patient safety by reducing the risk of misconnections between different delivery systems.
ISO 80369-5 is a section of the ISO 80369 series of international standards titled “Small-bore connectors for liquids and gases in healthcare applications” which specifically applies to limb cuff applications. “Small-bore” refers to the size of the opening of the connector, in this case less than 8.5 mm. This article delves into the relevance of ISO 80369-5, its impact, and the steps medical device companies are taking to adhere to this standard.